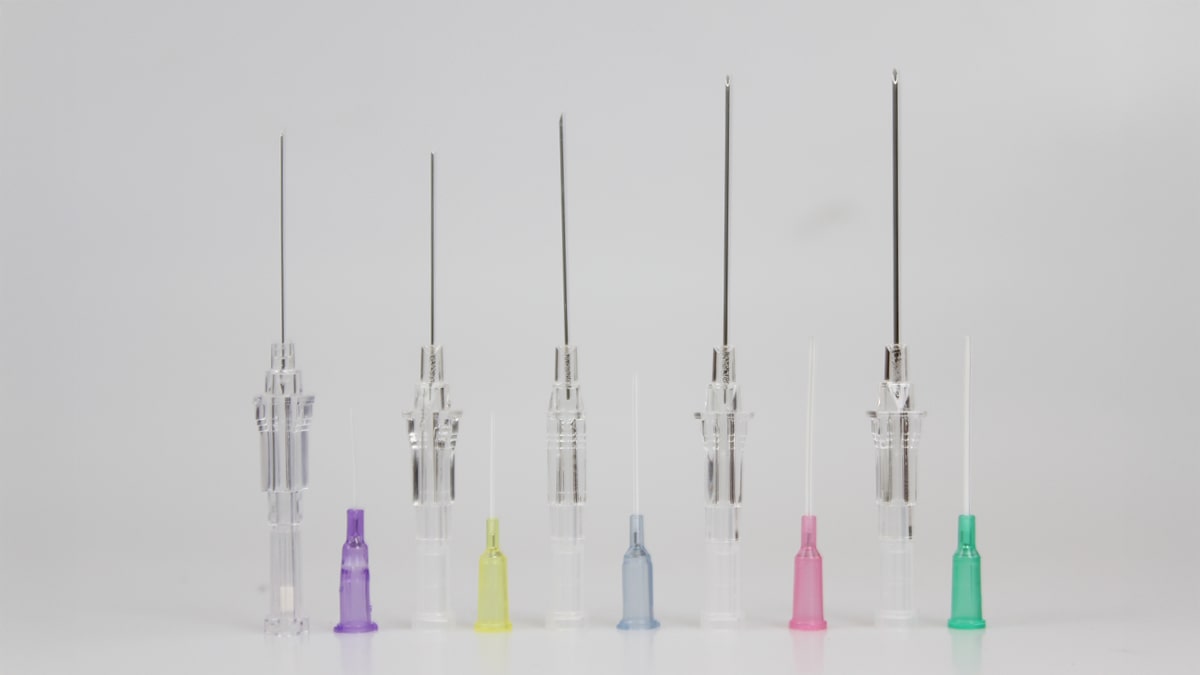
Drug delivery systems are among the most fundamental and critical components of modern healthcare, directly impacting patient safety and clinical practice. Top Point provides a comprehensive range of disposable medical devices for drug administration, including syringes, needles, IV catheters, IV administration sets, and blood transfusion sets, while prioritizing accuracy and safety at every stage of use.
Full-Cycle Medical Device Manufacturing Solutions
TOP-POINT supplies a full spectrum of high-quality, disposable medical products,
manufactured under strict ISO 13485 standards for safety and reliability.
High-speed automation for ISO-certified assembly.
Cost-effective, customized solutions for global partners.
Precision grinding technology for high-quality cannulas
End-to-end solutions: from feasibility to installation.
Follow our global footprint and commitment to innovation. This section features press releases,
exhibition participation, and key achievements that solidify our market leadership.
Establishing a disposable syringe manufacturing facility is a complex project that extends far beyond the purchase of production equipment. While many companies are drawn to the medical device industry by its stable demand and strong global growth potential, the practical challenges of setting up a manufacturing operation are often far more complex than initially anticipated.
Medical cannulas are tubular components used across a wide range of clinical procedures, including injections, catheterization, and endoscopy, where they play a critical role in delivering or removing fluids and medications. Custom cannulas, in particular, are manufactured beyond standard specifications to meet application-specific requirements such as precise outer and inner diameters (OD/ID), customized lengths, and controlled surface finishing.
In the medical device industry, selecting the right manufacturing partner is a critical decision that directly impacts product quality, operational reliability, and long-term market success. For companies targeting global markets, this choice carries even greater significance, as medical devices are inherently linked to patient safety and stringent regulatory requirements.
Young Corporation has been officially reappointed as a Designated Trading Company by the Ministry of Trade, Industry and Energy (MOTIE) of Korea. The 2025 Designation Ceremony, co-hosted by KOTRA and the Korea International Trade Association, was held on July 8, 2025, at COEX in Seoul.
Young Corporation successfully wrapped up its second participation in KIMES 2025, Korea’s leading international medical and hospital equipment exhibition held at COEX, Seoul. As one of the key players in the medical device field, Young Corporation once again drew attention with its innovative product lineup and customized solutions.
YOUNG CORPORATION: TOP-POINT is excited to announce its return to the 40th Korea International Medical & Hospital Equipment Show, KIMES 2025, held at COEX, Seoul. As a trusted leader in medical device manufacturing and automation, we invite you to Visit us at 1F, Hall B Lobby, Booth No. BL113. We will be showcasing our newest Innovative Medical Solutions, ranging from high-precision cannulas to advanced syringe manufacturing automation. Our sustained participation reaffirms our commitment to driving excellence and innovation in the global healthcare industry.
Young Corporation participated in MD&M WEST 2025, held in Anaheim, USA, marking its debut at this prestigious event. MD&M WEST is one of the world’s leading exhibitions for medical device manufacturing and design, offering a comprehensive view of the latest technologies and trends in the global medical device industry.
From June 19 to 21, 2024, Young Corporation attended the Florida International Medical Expo (FIME) held in Florida, USA, successfully gathering industry insights and conducting market research. FIME is the largest medical device exhibition in North America, bringing together medical device manufacturers and experts from around the globe.
Find quick answers to common questions about our Turnkey Solutions, OEM services, ISO 13485 compliance, and product quality.
If you don't see your question, please Contact Our Experts.
What is included in your Medical Device Turnkey Factory Setup?
Our turnkey solution is comprehensive, covering everything from initial factory design and layout to the supply and installation of full automation machinery, cleanroom validation, and technical training for operations.
Are your manufacturing processes compliant with international quality standards?
Yes, all our manufacturing and quality management processes are certified under ISO 13485:2016. This guarantees the highest standards of safety and efficacy for all our medical devices.
Do you provide OEM and Contract Manufacturing services?
Absolutely. We offer full OEM and contract manufacturing services for various disposable medical devices, utilizing our ISO 13485-certified production facilities to meet your specific design and volume requirements.
What types of medical devices do you specialize in manufacturing?
Our core expertise lies in the production of high-precision disposable items, including syringes, hypodermic needles, cannulas, and various IV administration and transfusion sets.
Can you support factory set up different countries and customer's requirements?
Yes. We have experience working in various countries across Asia, the Middle East, Africa, the United States, and other regions. We provide flexible, fully customized turnkey plant solutions tailored to the local conditions and specific requirements of customers in various countries.